CFAX12 for in vitro analysis of cigarette smoke effects on-chip
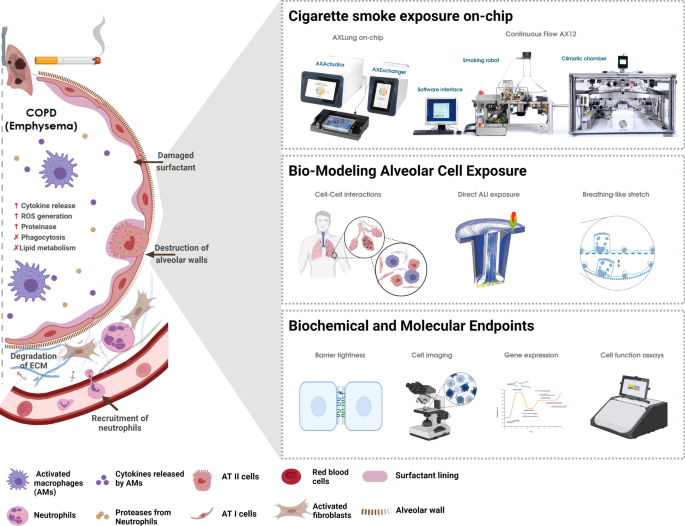
The Continuous Flow AX12 (CFAX12) exposure system was employed to investigate the effects of cigarette smoke on lung cells cultured in the AX12 Lung-on-chip (AlveoliX, Switzerland) (Fig. 2A). The system integrates a modified smoking machine (VC10, Vitrocell Systems, Germany) described earlier, which has been adopted for the current study to accommodate the AX12. Briefly, the smoke machine VC10 is connected to an exposure module housed within a climatic chamber, enabling precise control of environmental conditions during the experiments. The VC10 generates and delivers cigarette smoke to the exposure module. Cigarettes are automatically loaded from a magazine (Supplementary Fig. 1B) into the cigarette holder port (Fig. 2C, Supplementary Fig. 1F), where an electric lighter (Fig. 2D) ignited them. A syringe pump (Supplementary Fig. 1H), designed to mimic the inhalation process of a human smoker, drew puffs, and transferred the smoke to the dilution system (Supplementary Fig. 1C). The VC10 allows for adjustable puff parameters, including volume, duration, interval, and frequency. At the end of each smoking cycle, the butt extractor (Supplementary Fig. 1G) retracts all the burnt cigarettes and automatically dispenses them into an in-built ashtray. For this study, the Health Canada Intense (HCI) smoking regime was employed in terms of puff volume, duration, and number. Specifically, one 1R6F standard research cigarette (Kentucky Tobacco Research & Development Center, Lexington, KY, USA) was smoked per cycle (one cycle in total), with a puff volume of 55 mL, a puff duration of 2 s, a puff interval of 30 s, and a puff exhaust time of 8 s (Fig. 2B). A dilution airflow of 0.5L/min was applied, resulting in a dilution factor of 0.1803 (Dilution Factor = Volume of Smoke / (Volume of Smoke + Volume of Fresh Air). This standardized protocol facilitates the reproducibility and comparability of results. The exposure module was modified to house the AX12 lung-on-chip (Fig. 2E), which was maintained at a constant physiological temperature of 37 °C and humidity (~ 95%), achieved through the controlled environment of the climatic chamber assisted with additional humidification station (Supplementary Fig. 1A) attached with water baths (Supplementary Fig. 1D). The cigarette smoke generated by the VC10 smoking machine was diluted and homogenized with cigarette smoke-free air before being delivered to the exposure head. This setup ensures that the smoke reaches the cells cultured at the ALI condition in a controlled and uniform manner (Fig. 2G). The smoke delivery system included a series of flow controllers that regulated the air and smoke flow rates to achieve the desired dilution (Supplementary Fig. 1E). The exposure head, equipped with trumpet-shaped inlets, ensured even distribution of smoke over the cells. A vacuum pump generated a flow that directed the aerosol towards the cells, while the excess smoke was exhausted through an exhaust tube (Fig. 2F). The device was installed by the manufacturer and subsequently underwent Installation Qualification (IQ) and Performance Qualification (PQ) in accordance with their specifications at the time. Thereafter, performance was monitored daily through leakage tests and the assessment and adjustment of the puff volume. The CFAX12 system allowed for the precise adjustment of smoke concentration, simulating real-world smoking conditions. The exposure module featured two parallel aerosol channels, enabling simultaneous exposure to different conditions within the same experiment. Typically, one chip of the AX12 was exposed to cigarette smoke, while the other chip was exposed to clean air as a control. This setup provided a robust comparative analysis of smoke-induced effects versus healthy baseline conditions. Throughout the experiments, the system’s software interface recorded and monitored all relevant parameters, including puff volume, duration, interval, and frequency.

Components of the CFAX12 System. (A) Overview of the three main elements of the CFAX12 (from left to right): the computer system with the software interface, the VC10 smoking robot and the Climatic chamber. (B) The operation is controlled via a software interface where the Health Canada Intense (HCI) smoking regime was selected. (C) The cigarettes are loaded into the cigarette holders fully automatically and without damage, with the rotation of the holder controlled by a stepper motor. (D) The electric lighter to ensure automatic and contactless ignition. (E) The lung-on-chip consumable, AX12. (F) The VC10 smoking robot initiates the process delivering the smoke to the dilution system, where dilution air is added to the smoke feed stream. The whole smoke dilutes in the dilution module, passes through the exposure head, and reaches the AX12 with the cells seeded, secured in the exposure module. A sample flow generated by a vacuum pump (VP) diverts the aerosol towards the cells via “trumpet” inlets, and after contacting the cells, the smoke exits through an exhaust tube; the continuous dilution air flow ensures that any remaining smoke is also exhausted. (G).
Preparation of cigarette smoke extract
Cigarette smoke extract (CSE) was prepared utilizing the VC10 smoking robot component of the CFAX12. One 1R6F cigarette was loaded into the magazine which loaded it into the cigarette holder port. The smoke generated was drawn by a syringe pump, mimicking smoker inhalation, and delivered to a T-tube connected to a PBS-filled (10 ml) falcon tube. Smoke was bubbled through the PBS to create fresh 100% CSE extract before each experimental round. After completing the smoking cycle, the CSE was filtered using a 0.22 µm sterile filter and diluted with warm culture medium to the desired concentration (0%, 2.5%, 5%, 10%, 25%, 50% and 100%). The prepared CSE was then applied to the apical side of the cell cultures in the AX12 for subsequent experimental procedures.
AXLung on-chip technology
As described previously35,36, the AXLung-on-chip system (AlveoliX AG, Switzerland) comprises the Lung-on-chip (AX12) consumable (Fig. 2B), two electro-pneumatic devices (AXExchanger and AXBreather), and the interface platform (AXDock) (Fig. 1). Cell seeding and medium exchanges were performed according to the manufacturer’s recommendations (AlveoliX AG). To initiate the breathing mechanism, the AX12 was positioned within the AXDock inside the incubator. The three-dimensional (3D) cyclic stretch (10% linear strain, 0.2 Hz) was then initiated via a touchscreen control on the AXBreather, which deflected the microdiaphragm by generating negative cyclic pressure. Both stretch (Str) and non-stretched static conditions were run simultaneously on a single AX12, as the two chips were controlled independently.
Cell culture and treatment
The alveolar epithelial cell line (AXiAECs) used in this study was procured from AlveoliX (Switzerland). These cells were derived from primary human alveolar epithelial cells (AECs) isolated from resected lung tissue and subsequently immortalized using InscreeneX’s CI-Screen technology, as described by Lipps et al.37. The AXiAECs were cultured according to the manufacturer’s instructions, as previously described and characterized by Sengupta et al.35.
Human Lung Microvascular Endothelial Cells (hLMVECs) purchased from PromoCell were expanded in flasks using AX Endothelial Medium. hLMVECs were first seeded on the basolateral side of each membrane in the AX12 at a density of 106,000cells/cm2, followed by a 2-h incubation, and then AXiAECs were seeded apically at a density of 409,000 cells/cm2 on day 0 for all co-culture experiments. The cells were maintained at 37 °C, 5% CO2, with the medium replaced every 2 days. On the AX12, AXiAECs were maintained in AX Alveolar Epithelial Barrier Medium (AlveoliX, Switzerland) supplemented with 1% penicillin–streptomycin (Thermo Fisher Scientific, Switzerland) for mono-culture studies and AXiAECs/hLMVECs were maintained in AX E2 Alveolar Barrier Medium (provided by AlveoliX) for all dual-culture studies. In all studies, the AXiAECs alone or in co-culture with the hLMVECs were maintained and differentiated on the AX12 for three weeks prior to cigarette smoke treatment or exposure conditions, where ALI and stretch was initiated in specified experiments around 10 days prior to smoke exposure.
Human primary blood-derived monocytes (pBDMCs) were isolated from donor buffy coats using the EasySep™ Direct Human Monocyte Isolation Kit (Stemcell, #19669), which isolates primary CD14 + monocytes via negative immunomagnetic selection according to the manufacturer’s protocol (Supplementary Fig. 2A). The pBDMCs were cultured in RPMI medium (21875-034, Gibco) with 10% FBS, 1% P/S, and 2 mM L-Glutamine for one day before being differentiated into macrophages (primary blood-derived macrophages; pBDMs) in the same medium supplemented with 50 ng/mL M-CSF (Peprotech, #300-25) for six days, with medium exchanges every 2 days. After six days, the cells were washed with PBS and detached using pre-warmed Accutase (Sigma Aldrich, # A6964). The pBDMs cell suspension was centrifuged, and the cell pellet was resuspended in fresh triple-culture medium (2 parts of AX E2 Alveolar Barrier Medium: 1part of RPMI) and added on the apical side at a ratio of 1:7 (pBDMs: AXiAECs). The usage of donor-derived pBDMCs was approved by the Blutspendezentrum (SRK Bern).
The THP-1 monocytes (ATCC; TIB-202) are an immortalized monocyte-like cell line derived from the peripheral blood of a patient with acute monocytic leukemia. These cells were cultured in RPMI medium (21875-034, Gibco) supplemented with 10% FBS, 1% P/S, and 2 mM L-Glutamine. Differentiation of THP-1 monocytes into macrophage-like cells was induced using 100 nM phorbol 12-myristate 13-acetate (PMA; Sigma) and incubated for 48 h following the established protocol by Kletting et al.38. After incubation, the differentiated THP-1 macrophages were allowed to rest in PMA-free medium for an additional 24 h prior to seeding in the AX12. For triple-culture experiments the THP1s were added apically at a ratio of 1:7 (THP-1: AXiAECs).
For all experiments conducted, unless specified otherwise, 0 h refers to the time point immediately before exposure to CS or CSE. The 4 h, 24 h, and 48 h time points indicate the number of hours post-exposure.
Both pBDMs and THP-1 macrophages were added to the epithelial/endothelial (AXiAECs/hLMVECs) co-culture 5 days prior to CS exposure. All cell lines were routinely monitored for mycoplasma contamination using a mycoplasma detection kit (MycoStrip, InvivoGen).
Pulmonary surfactant
Porcine-derived pulmonary surfactant was added to the co-culture conditions 24 h before smoke exposure (Fig. 7A). Curosurf® (poractant-alpha, Chiesi Farmaceutici, Parma, Italy) is a clinical surfactant preparation provided as a suspension at 80 mg/mL containing more than 90% of surfactant phospholipids and around 1% by mass of surfactant proteins SP-B and SP-C. It was carefully diluted in 1X PBS (Sigma Aldrich, #D8537) to a concentration of 30 µg/µl and 5 µl was added on the apical side of the membrane per well in the AX12.
Flow cytometry analysis
Freshly isolated primary blood-derived monocytes (pBDMCs) were analyzed using flow cytometry. In brief, cells were incubated at 4 °C with the following antibodies: anti-CD45 (Biolegend, # 30-F11), anti-CD14 (Biolegend, # M5E2), as well as with Live Dead Fixable Blue (Thermofisher) for live/dead cell discrimination. After washing, samples were acquired using a LSRII SORP device (BD Bioscience, USA) and analyzed with the FlowJo software (version 10.8.1, BD Bioscience).
Liquid chromatography–mass spectrometry measurements
CS exposure was performed in the CFAX12, and smoke was trapped in 1 × PBS filled wells in the AX12. Nicotine, cotinine and 3’-hydroxy-cotinine (OH-cotinine) were measured in PBS using a quantitative liquid chromatography coupled to tandem mass spectrometry (LC–MS/MS) method, based on a previously validated method for saliva (as established in39). In brief, the measurements were performed using a Shimadzu Prominence HPLC (Shimadzu, Reinach, Switzerland) coupled to a SCIEX 4000 QTrap mass spectrometer (AB Sciex, Darmstadt, Germany) and a PAL autosampler (CTC Analytics, Zwingen, Switzerland). Chromatographical separation was achieved with an XBridge BEH C18 column (3.5 µm, 4.6 × 100 mm, 130 Å, Waters, Dättwil, Switzerland) and a delay column of the same specifications was used to minimize environmental contamination. Mobile phase A consisted of 0.01% NH4OH in water and mobile phase B of 0.01% NH4OH in methanol with a gradient starting at 5% mobile phase B which linearly increased to 90% B at 2 min and to 100% B at 2.5 min. The PBS samples were combined with 80% methanol containing the deuterated internal standards (IS) (25 μL sample + 250 μL IS mix). For quantitation, calibration curves were constructed from at least six consecutive calibrators in the range from 1 to 4000 ng/ml, covering the relevant concentration ranges in the samples. When necessary, the samples were pre-diluted 10 × with 80% MeOH to ensure concentrations within the dynamic range of the method. The lower limits of quantification (LLOQs) of the method were 2, 1 and 1 ng/mL for nicotine, cotinine and 3’-OH-cotinine, respectively.
TER measurement
To determine barrier formation, trans-barrier electrical resistance (TER) measurements were taken every two days, starting 48 h after cell seeding as discussed previously35. In brief, a commercially available 96-well plate electrode (STX100MC96, World Precision Instruments) and an Epithelial Volt/Ohm Meter (EVOM3; World Precision Instruments) was used. TER was measured in mono, and co-culture conditions until endpoint. Before taking a reading, the electrodes were carefully sterilized using 70% v/v ethanol for 5 min and then rinsed in distilled water for another 5 min at room temperature (RT). The “TER measurement” option was initiated on the AXExchanger while taking readings. For cells kept in ALI, pre-warmed 1X sterile PBS was added 15 min prior to the measurements and then later re-equilibrated after TER readings. The background TER (Ohm) data was obtained from a well having no cells. For analysis, the background subtracted TER (Ohm) values were multiplied by the surface area of each cell culture well (0.071 cm2 in AX12) to obtain the final TER reading in Ohm.cm2.
qRT-PCR
Total RNA was isolated separately from the apical and basal chamber and subsequently purified using the Direct-zol™ RNA Microprep kit (Zymo Research, Switzerland) using the manufacturer protocol. Purity and concentration of RNA was analyzed using a Nano-Drop Spectrophotometer (Thermo Fischer Scientific, Switzerland). Next, cDNA was transcribed using the Super Script III Reverse Transcriptase kit (Life Technologies, Switzerland). Finally, qRT-PCR reactions were performed in triplicates with SYBR® Select Master Mix (Thermo Scientific) in an ABI7500 Fast (Applied Biosystems) real-time qRT-PCR system. Target gene expression was normalized to housekeeping gene expression (GAPDH). The primer sequences are provided in supplementary Table 1.
ELISA
IL8 ELISA was performed with a human IL8 ELISA kit (BioLegend, #431504), according to the manufacturer’s instructions. All ELISA experiments were repeated in technical duplicates with apical supernatants collected from the AX12 Lung-on-chip.
LDH cytotoxicity assay
Collected apical supernatants from individual AX12 wells were analyzed for Lactate Dehydrogenase (LDH) release. The LDH-Glo cytotoxicity assay kit (Promega, #J2381) was used according to the manufacturer’s protocol. All LDH experiments were repeated in technical duplicates. The respective cell culture medium was taken as the “blank” control. Healthy untreated cells were considered as “negative” control for the assay, whereas cells treated with 1% Triton X-100 were considered as the “positive” control for maximum LDH release. Luminescence was measured using a microplate reader (Tecan Reader M1000).
Measurement of ROS activity
To measure reactive oxygen species (ROS) from smoke exposed cells, the DCFDA/H2DCFDA cellular ROS assay kit (Abcam; ab113851) was used according to the manufacturer’s instructions. A blank or negative control (well with no cells) was included to normalize the background for subtraction. A positive maximal ROS control was prepared with Ter-butyl hydrogen peroxide (TBHP) at a concentration of 150 μM in buffer (provided within the kit). DCF amount obtained from the cells was then finally detected at Ex/Em = 485/535 nm in a fluorescence microplate reader (Tecan Reader M1000).
Immunofluorescence staining and imaging
Cells in AX12 were fixed using 4% paraformaldehyde in distilled PBS- (PBS without calcium and magnesium). “Medium exchange” function on the AXExchanger was used to fix the cells within the basal chamber. Chips from the AX12 were unscrewed and opened using the AXDisassembly tool (AlveoliX, Switzerland) prior to staining and mounting. Cells were first permeabilized using 0.1% Triton X-100 (Sigma-Aldrich, Germany) dissolved in PBS for 15 min at RT and then blocking buffer (2% BSA in sterile PBS) was added. The cells were incubated for 1 h at RT with the blocking buffer. The antibodies used for the study are conjugated Alexa Fluor 488 anti-ZO1 (Thermo Fischer Scientific, #339188) and conjugated Alexa Fluor 568 anti-CD31/Pecam-1 (Cell Signaling; #61255). The antibodies were diluted in 2% BSA/PBS and incubated overnight at 4 °C. Nuclei were stained with Hoechst (1:1000 dilution; Invitrogen, #33342). The actin cytoskeleton was visualized using the conjugated Alexa Fluor 647 phalloidin stain (1:300 dilution; # PHDN1-A, Cytoskeleton, Inc.). Lastly, the stained membranes were sealed between two glass coverslips using a mounting medium (Sigma-Aldrich, #F6182). Images were obtained using a confocal laser scanning microscope (Zeiss LSM 980) using appropriate settings.
Statistical analysis
All bar graph data are presented as mean ± standard error of the mean (SEM) unless specified otherwise. For all AX12 experiments, “N” represents the experimental repetitions, and “n” represents the number of individual wells estimated for all experiments with mRNA and cell supernatants. All statistical tests were performed using t-tests, one-way or two-way analysis of variance (ANOVA) with p values adjusted for multiple comparisons using Tukey’s multiple comparisons test, *p < 0.0332, **p < 0.0021, ***p < 0.0002, ****p < 0.0001. For all studies, the absence of statistical significance brackets indicated that the p-value was greater than 0.0332. GraphPad Prism software v10.0 was used for data analysis. The exact number of repeats performed for each experiment is indicated in the corresponding figure legends.
link